12 gram CO2 cartridge

A 12 gram CO2 cartridge, known commercially as a Powerlet, contains 12 grams of CO2 in a liquid/gas equilibrium. The cartridge is made of thick steel with a thin welded cap. When the cap is pierced, the CO2 rapidly expands. Cartridges can be purchased in bulk for around fifty cents each. These cartridges offer a portable, convenient method of pressurizing a pneumatic cannon. Because CO2 operates at extreme pressures, these cartridges must be treated with appropriate caution.
Usage

The simplest way to connect a 12 gram CO2 cartridge is with a quick-change adapter. This type of adapter is very common, and can be found for $10-$20 on eBay. It is intended for use in paintball, allowing a paintball marker to draw power from a 12 gram CO2 cartridge instead of a remote connection.
A full CO2 cartridge is placed in the adapter's shell. The shell is screwed tightly against a pin. When the cartridge is pierced, CO2 will rapidly expand forward through the adapter. Generally, each CO2 cartridge is single-use.
Connecting
Quick-change adapters have male "ASA" threads. ASA is a paintball-specific thread size that is nearly identical to 1/2" NPT threading. However, ASA threads are parallel and rely on an O-ring for an airtight seal whereas NPT threads are tapered and thus seal automatically.

Male ASA threads will form an airtight seal with female 1/2" NPT threads, given sufficient tightening and liberal use of teflon tape. A more proper method involves connecting the quick-change adapter to a CO2 remote on/off adapter, which has a female 1/8" NPT thread. (The on/off functionality will not work.)
Dimensions
The cartridges have a diameter of 49/64" and a total length of 3.25". Each cartridge has a mass of 31.5 grams empty, and 43.5 grams when full. The cartridge has an internal volume of precisely 14 cubic centimeters.
Pressure
Below 87.8°F, a 12 gram CO2 cartridge contains a mixture of liquid and gaseous CO2 in equilibrium. When the gaseous CO2 expands, some of the liquid CO2 immediately boils to take its place. As long as liquid remains, the operating pressure will be the vapor pressure of CO2 at the current temperature (usually 800+ PSI).
Above 87.8°F, or if the CO2 is given enough room to expand, the CO2 will be purely gas. It will behave like an ideal gas, so the operating pressure can be calculated using the Ideal Gas Law, given the volume to be filled and the ambient temperature. For this calculation, note that 12 grams of CO2 is roughly 0.273 moles of CO2.
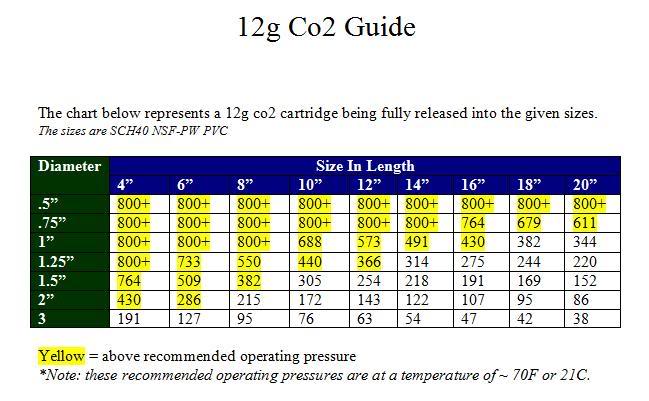
At 70°F, the following approximation (derived from the Ideal Gas Law) indicates the pressure of a given chamber when filled with a 12g CO2 cartridge:
Pressure (psi) = 5910 / Volume (in^3)
Remember that below 87.8°F, CO2's pressure is limited by its vapor pressure.
Cooling
When released from a 12 gram cartridge, CO2 will rapidly cool everything it flows through. Extra precautions should be taken when using 12 gram cartridges with temperature-sensitive materials such as PVC, which becomes brittle when cold.
Other uses
12 gram CO2 cartridges can be used effectively as projectiles. They are dense, consistently sized, and fit well in 3/4" SCH 40 PVC.
See also
The CO2 Cartridge...an Under-Appreciated Marvel of Technology!