Butane
Jump to navigation
Jump to search
Butane is quite similar to propane, but with a slightly lower stoichiometric value of 3.23%. Pure butane is found in disposable lighters and lighter refill canisters.
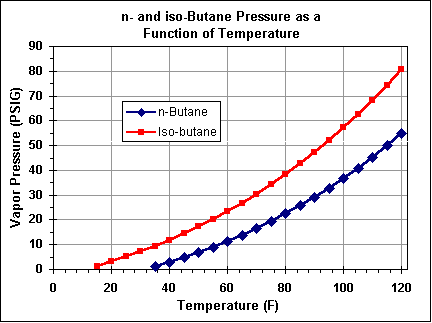
Butane has a lower vapor pressure than propane, and its boiling point is -0.5°C (31.1°F), limiting it's use to higher temperature environments. It is most easily used the same way as aerosol fuels.
Disposable canisters with mixtures of butane, propane and (sometimes) isobutane are sold for camping gear and blowtorches, this gas evaporates at lower temperatures and can be used in cold weather.
Molecular Formula: C4H10
n-Butane Extended Formula: CH3CH2CH2CH3
iso-Butane Extended Formula: CH3CH(CH3)CH3
Combustion: 2C4H10 + 13O2 ==> 8CO2 + 10H2O